
Natal conditions may influence susceptibility.

Natal conditions may influence susceptibility.

Lack of trust in vaccines abetted by online articles and past wrongdoing by the drug industry.
First non-insulin drug for pediatric use approved in nearly 20 years.
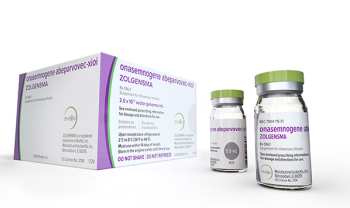
Zolgensma (onasemnogene abeparvovec-xioi) is an adeno-associated virus vector-based gene therapy
North Carolina working to expand the number of vaccines pharmacists can administer.
HZ in children is uncommon and is 10 times less common in kids who’ve been vaccinated against chickenpox.

Anthelmintic tablet for patients 6 years of age and older.

Each year, approximately 200,000 children ages 17 years and younger land in emergency departments (EDs) from adverse drug events (ADEs). Children younger than 5 years of age are most likely to visit the ED for ADEs, according to the CDC.

A study of vaccination exemptions in kids found that some doctors are charging anti-vaccine parents for them.

Continuing to work with FDA to ease the ongoing shortage of injectable methotrexate, APP Pharmaceuticals this week released another 10,000 vials of preservative-free methotrexate (Methotrexate Injection), a drug used in the treatment of pediatric oncology and hematology

FDA has approved lucinactant (Surfaxin, Discovery Labs) for the prevention of respiratory distress syndrome (RDS) in premature infants at high risk for RDS. Surfaxin is the first synthetic, peptide-containing surfactant approved for use in neonatal medicine.

New guidelines for the use of the quadrivalent human papillomavirus vaccine, published by the American Academy of Pediatrics, recommend for the first time vaccinating adolescent boys as well as girls.

Zinc used in addition to antibiotics significantly reduced mortality in children ages 6 months to 59 months with severe pneumonia when compared with antibiotics alone, according to the findings of a recent study done in Uganda.

More than a half million bottles of Infants' Tylenol Oral Suspension, 1 oz. grape, have been recalled voluntarily by McNeil Consumer Healthcare because of complaints about using the dosing system, the company announced.

FDA and 3 drug manufacturers say they are working to allay the concerns of health organizations that thesupply of methotrexate -- used to treat acute lymphoblastic leukemia in children – is in danger of running out

Commercial electronic prescribing systems can significantly reduce medication errors in the hospital setting, according to a study published in PLoS Medicine in January

FDA recently approved methylphenidate hydrochloride extended-release capsules (LA) CII, USP, the generic equivalent of Ritalin LA. Actavis Group, an international generic pharmaceuticals company, received first-to-file exclusivity on the 20-mg, 30-mg, and 40-mg strengths.

Children who are overweight or obese have 3 times as much risk of high blood pressure as children of normal weight have, reported a study published recently in Hypertension: Journal of the American Heart Association.

Annual flu vaccines are the best way to protect children from life-threatening pneumonia, according to new guidelines from the Pediatric Infectious Diseases Society and the Infectious Disease Society of America.

Only one-fifth of children in Ohio who are prescribed antidepressants complete the recommended mimimum six-month course of drugs to treat depression, a new study reported.

FDA has approved 2 new strengths (11.25 mg and 30 mg) for 3-month administration of leuprolide acetate for depot suspension (Lupron Depot-PED, Abbott) for the treatment of children with central precocious puberty.

FDA approved the use of meningococcal (A, C, Y, W-135) polysaccharide diphtheria toxoid conjugate vaccine (Menactra, Sanofi Pasteur) in children aged as young as 9 months for the prevention of invasive meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y, and W-135.

The American College of Rheumatology has developed new guidelines for starting and monitoring treatments for children with juvenile idiopathic arthritis.

SmartTots, a partnership between the International Anesthesia Research Society and FDA, is gathering more research on safe anesthesia for children under 4 years old.