Influenza
Latest News
Latest Videos
CME Content
More News
Secretary of US Health and Human Services Robert F. Kennedy Jr also approved all recommendations issued from the April ACIP meeting.
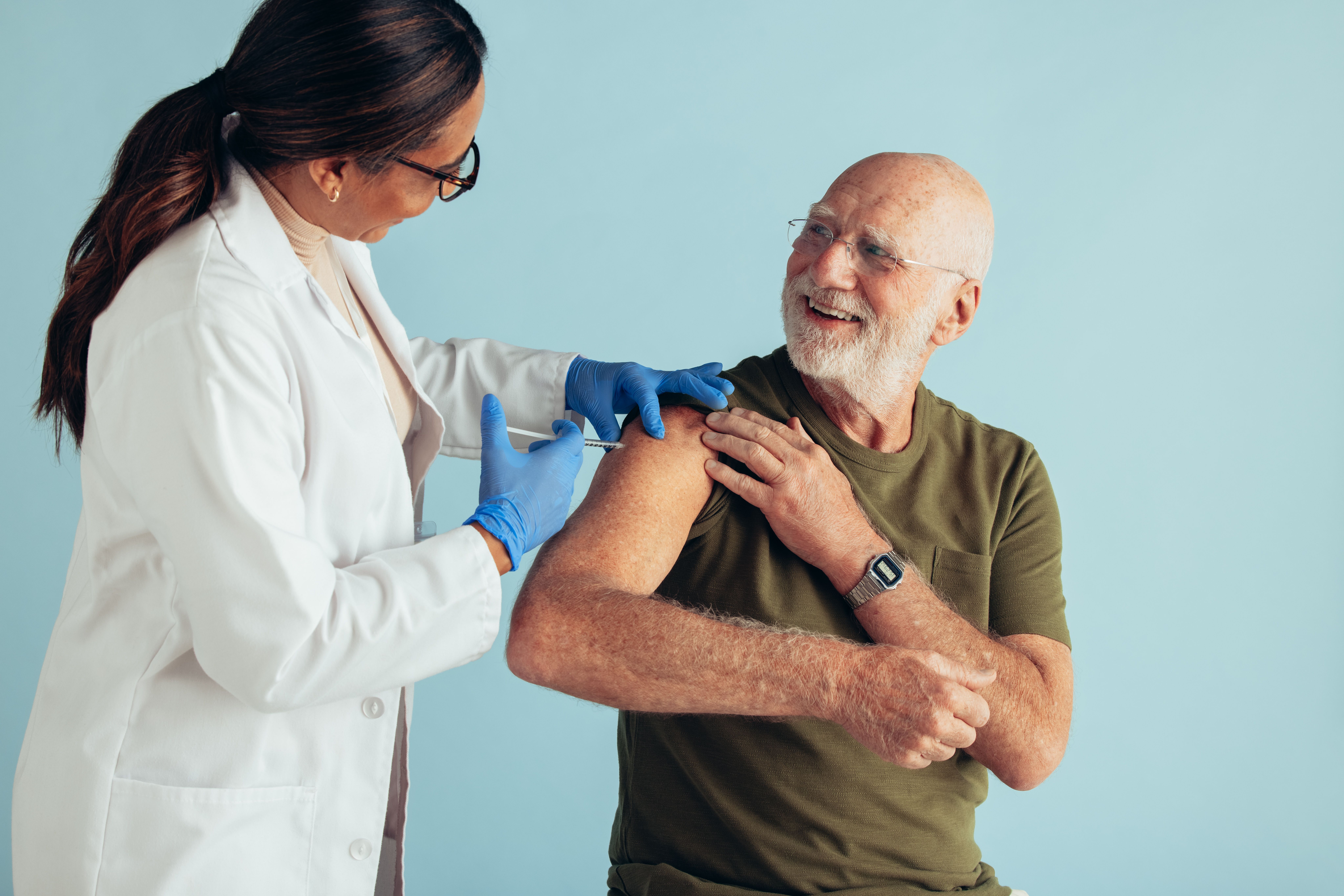
Flu vaccination significantly reduces illness in vaccinated and unvaccinated individuals, highlighting its importance in public health strategies.

Researchers determined the impact COVID-19 interventions had on Danish children’s incidence of RSV, influenza, and invasive pneumococcal disease.
ACIP also discusses COVID-19 epidemiology as well as safety and efficacy for the COVID-19 vaccines.

CD388 is being investigated as a prevention method for seasonal influenza in unvaccinated patients aged 18 to 64 years.
Researchers wanted to address the potential impact of pharmacist-led influenza immunization services on vaccine uptake in older adults living in Denmark.
Currently under development, the manufacturer’s investigational pandemic influenza vaccine is designed to target the H5 avian influenza virus, or bird flu.
The investigators hope to pursue a human vaccine in order to improve upon the already existing vaccines.

Patients with psychosis, depression, and anxiety exhibit higher rates of vaccine hesitancy.

Global virus and public health experts gathered to assess the need for an enhanced response to the bird flu spreading among humans and animals.
Investigators specifically analyze information on influenza and pneumococcal vaccinations.
The designation addresses the unmet need for prevention of H5N1, which remains a global health risk, and a phase 1 trial of the vaccine was initiated in November 2024.

Coalition for Epidemic Preparedness Innovations experts warned of the potential for bird flu to become a pandemic event if officials don’t prepare quickly.
Researchers investigated the acceptability of community pharmacy-led influenza vaccination and patients’ willingness to return for future immunization.
With the disease burden remaining high, CVS Health announces new flu testing and treatment capabilities at approximately 1600 locations in selected states.

US Secretary of Agriculture Brooke Rollins detailed her organization’s plans to combat the bird flu and quell the concerns of rising grocery store costs.

The next-gen molecular Metrix was given emergency use authorization for its ability to test for SARS-CoV-2 and influenza A and B viruses.

Experts believe a bird flu outbreak is responsible for hundreds of dead birds washing ashore on the beaches of Chicago.
In their exploration of 2 separate respiratory virus seasons, researchers examined the severity of illness for SARS-CoV-2, influenza, and respiratory syncytial virus.
The US government is entrusting mRNA vaccine pioneers Moderna to develop protection for the gradually increasing threat of avian influenza.
Researchers compared the safety and reactogenicity of simultaneous doses of recombinant zoster vaccine paired with 2 separate types of inactivated influenza vaccines.

There are a lack of data available on the optimal antiviral medication for nonsevere flu.

The individual who first reported severe bird flu and hospitalization has since succumbed to their illness.
Researchers compared simultaneous and sequential administration of the mRNA COVID-19 and inactivated influenza vaccines.
Using the DASH Rapid PCR System, providers can yield test results in under 1 minute of hands-on time.