Infectious Disease
Latest News
Latest Videos
CME Content
More News

Multifaceted antimicrobial stewardship initiatives significantly enhance antibiotic prescribing practices for respiratory infections, improving patient outcomes.

In an exploration of the National Retail Data Monitor, researchers determine how OTC sales contributed to disease surveillance.

From verifying vaccination status to administering immunizations, pharmacists can mitigate the spread of various infectious diseases.
Amid recent measles outbreaks in Texas and Ohio, researchers aim to understand MMR vaccine coverage following postelimination outbreaks throughout the country.

Among older adults with Medicare, researchers investigated national influenza vaccination patterns by race, education level, and rurality.
Researchers explored the impact of PCV10 on the distribution of invasive pneumococcal disease serotypes among patients in Lithuania.

Among participants in the CATCH trial, researchers explored the relationship between HPV vaccination and incidence of infection.

Searching for the clinical and economic impacts, researchers explored the implications of pharmacist-led anti-infective consultations.
FDA approves Biktarvy for HIV treatment in patients not virologically suppressed, enhancing options for those restarting antiretroviral therapy.

The decision makes the therapy the first and only twice-yearly option available in the United States for PrEP.

The therapy is the first and only oral direct acting antiviral approved by the FDA for HCV infection.

The therapy is the first and only RSV preventative option using the same dose regardless of patient weight.
Building trust with patients through fact-based information could help boost vaccination rates to the desirable coverage.
Pharmacy team members can help patients prepare for upcoming travels, ensuring they are up to date with the recommended vaccines for their locations.
Pharmacists play a critical role in vaccination and education for patients about the evolving infectious disease landscape.

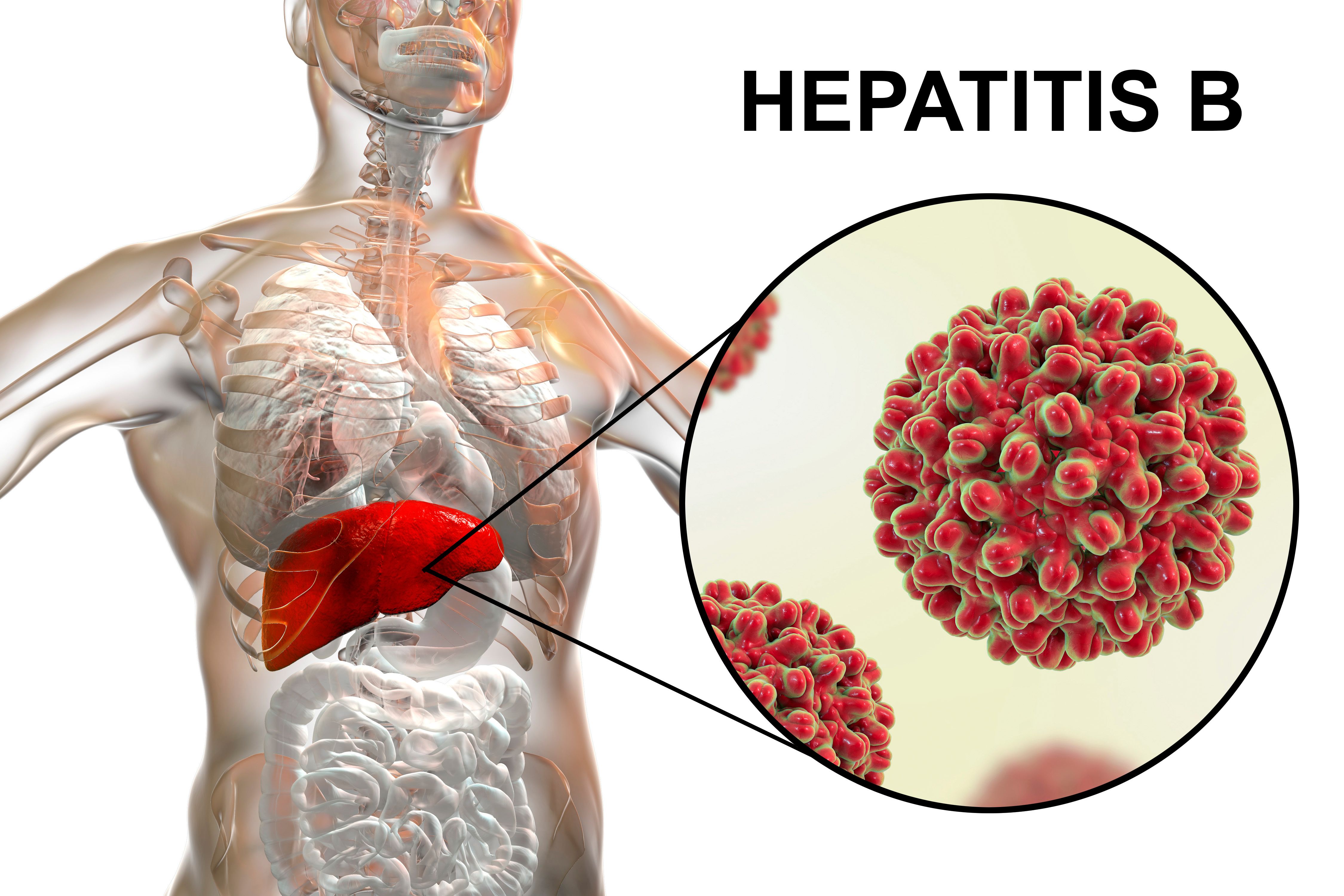
During Viral Hepatitis Awareness Month, Jeff Goad, PharmD, MPH, joined Over the Counter to discuss hepatitis vaccines and the common trends pharmacists in this space should keep in mind.
Approximately 67% of adults in the US know that vaccines do not cause autism in children, declining significantly from 74% in 2021.
Jeff Goad, PharmD, MPH, discusses hesitancy for hepatitis vaccines and the necessary advice pharmacists can utilize to help patients manage these misconceptions.
Jeff Goad, PharmD, MPH, discusses hepatitis viruses and the current vaccination schedules available for patients to stay protected.
The investigators hope to pursue a human vaccine in order to improve upon the already existing vaccines.
A phase 2 study found 95% of patients achieved a sustained virologic response at 12 weeks regardless of treatment adherence.

Global virus and public health experts gathered to assess the need for an enhanced response to the bird flu spreading among humans and animals.

The findings support the need for improved vaccination coverage to prevent the resurgence of previously eliminated infectious diseases in the United States.
In a new survey, approximately one-third of respondents have heard that the MMR vaccine is more dangerous than being infected with the infection.

Crystal Hodge discusses being transparent about adverse effects of the MMR vaccine and educating patients about measles.