Shingrix’s Efficacy Supported by Real-world Data
Study reports recombinant zoster vaccine is more than 85% effective overall.
The results of a recently published study concluded that the effectiveness of the recombinant zoster vaccine (RZV) (
RZV’s effectiveness in general practice had not been investigated previously to the new study. “Understanding the effectiveness of RZV in practice, outside of clinical trial settings, is crucial given the differences between clinical trial and general practice settings,” noted Yuwei Sun, MS, one of the study’s authors and an analyst with the University of California, San Francisco’s Francis I. Proctor Foundation for Research in Ophthalmology. “The clinical trials had standardized protocols for vaccine storage, administration, and herpes zoster diagnosis. In general practice, patients often have more health comorbidities.”
The findings, reported in
The retrospective cohort study looked at 4.7 million non-immunocompromised, vaccine age-eligible individuals from a deidentified claims database. A total of 173,745 adults were given 2 doses of RZV.
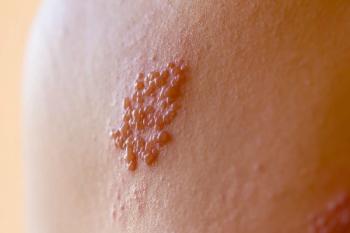
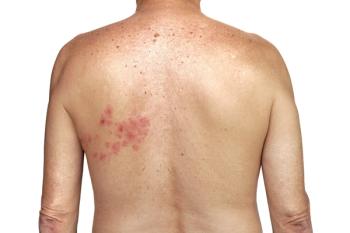
The results revealed that the HZ incidence rate was 258.8 cases per 100,000 person-years among those who received the vaccine compared with 893.1 per 100,000 person-years among the unvaccinated individuals.
RZV’s overall effectiveness was calculated to be 85.5%. Among patients aged 50 to 79 years, the rate was 86.8%, and 80.3% among patients aged 80 years or older. Among patients with a history of live zoster vaccine within 5 years of their inclusion in the study, the effective rate was 84.8%.
“Whenever you transition from a clinical trial to the real world, you wonder whether the results achieved in the clinical trial—in this case, excellent results—translate comparably to a real-world setting,” said William Schaffner, MD, medical director at the National Foundation for Infectious Diseases, in a prepared
According to the
RZV is the only vaccine against shingles currently available in the United States. In clinical trials, it was not associated with serious adverse events. The short-term
Individuals who have had shingles in the past can be vaccinated with RZV to help prevent future occurrences of the disease. There is no specific wait time indicated between having shingles and getting vaccinated, but it is recommended that the shingles rash has totally healed before getting the vaccine.
References
1. Sun Y, Kim E, Kong CL. Effectiveness of the recombinant zoster vaccine in adults aged 50 and older in the United States: a claims-based cohort study. Clinical Infectious Diseases. 2021.
2. Dreisbach EN. Real-world data trumpet effectiveness of recombinant zoster vaccine. Published February 27, 2021. Accessed March 31, 2021.