- Drug Topics May 2022
- Volume 166
- Issue 05
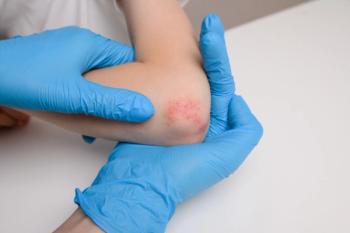
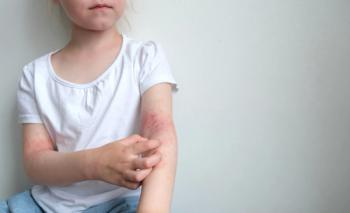
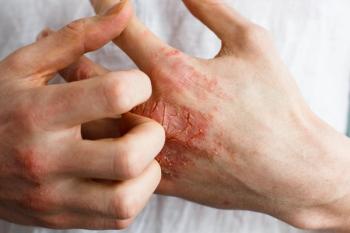
Abrocitinib for Atopic Dermatitis
The drug’s approval was based on the results of 3 clinical trials.
On January 14, 2022, the FDA approved abrocitinib (Cibinqo) for the treat- ment of adults with refractory, moderate to severe atopic dermatitis (AD) not adequately controlled with other systemic drug products, including biologics, or for patients in whom the use of those therapies is inadvisable.1 Abrocitinib is a Janus kinase inhibitor and is not recommended for use in combination with other Janus kinase inhibitors, biologic immunomodulators, or other immunosuppressants. Abrocitinib is approved at doses of 50 mg, 100 mg, and 200 mg.
Efficacy
Abrocitinib was approved based on the results of 3 randomized, double-blind, placebo-controlled studies: Trial-AD-1 (NCT03349060; n = 387; abrocitinib monotherapy), Trial-AD-2 (NCT03575871; n = 391, monotherapy), and Trial-AD-3 (NCT03720470; n = 837; combination therapy). In the 2 monotherapy trials, participants 12 years and older were randomly assigned to either placebo, 100 mg, or 200 mg groups. In the combination therapy trial, adult participants were randomly assigned to receive either oral or injectable placebo, abrocitinib 100 mg or 200 mg, or dupilumab (Dupixent) 300 mg. Abrocitinib efficacy was compared with dupilumab in terms of pruritus relief at 2 weeks. All participants in this trial also received background topical corticosteroids.2
In all 3 studies, the 12-week coprimary outcome measures were the percentage of participants achieving Investigator’s Global Assessment (IGA) response of clear (0) or almost clear (1) and a reduction of greater than or equal to 2 points from baseline and the percentage of participants achieving Eczema Area and Severity Index response of greater than or equal to 75% improvement. At the end of the 12-week period, both abrocitinib doses met the IGA end point (IGA, 0 or 1) vs placebo.1
Safety
The most common adverse events reported in greater than or equal to 5% of patients in the abrocitinib 100 mg, abrocitinib 200 mg, and placebo groups, respectively, included nasopharyngitis (12.4%, 8.7%, 7.9%), nausea (6%, 14.5%, 2.1%), and headache (6%, 7.8%, 3.5%).2
Abrocitinib prescribing information contains boxed warnings for increased risk of serious bacterial, fungal, viral, or opportunistic infections, including tuberculosis, leading to hospitalization or death. Additionally, major cardiac adverse events, malignancies, and thromboses have occurred with abrocitinib therapy in clinical studies for AD.2
Dosing
The recommended abrocitinib dose is 100 mg once daily, administered orally. In patients who do not respond to the 100-mg dose, dosing may be increased to 200 mg once daily. A 50-mg dose is approved for patients with moderate renal impairment (estimated glomerular filtration rate, 30-59 mL/min), for patients who are known or suspected to be poor metabolizers of cytochrome P450 (CYP) 2C19, or for patients receiving inhibitors of CYP2C19.2
Hooman Kashi is a PharmD candidate in the class of 2022 University of Connecticut School of Pharmacy, as well as a UConn Pharmacy Ambassador and UConn Leader’s Track Scholar. Kevin Ho is a PharmD candidate in the class of 2022 University of Connecticut School of Pharmacy, as well as a UConn Leader’s Track Scholar.
References
- U.S. FDA Approves Pfizer’s Cibinqo (abrocitinib) for adults with moderate-to-severe atopic dermatitis. News release. Pfizer Inc. January 14, 2022. Accessed April 13, 2022.
https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-pfizers-cibinqor-abrocitinib-adults - Cibinqo. Prescribing information. Pfizer Inc; 2022. Accessed April 13, 2022.
https://cdn.pfizer.com/pfizercom/USPI_Med_Guide_CIBINQO_Abrocitinib_tablet.pdf
Articles in this issue
over 3 years ago
The Future of Pharmacy is Digitalover 3 years ago
Pharmacy’s New "Dirty Little F-Word”over 3 years ago
How to: A Guide for Launching HbA1c Point-of-Care Testingalmost 4 years ago
Community Pharmacies Make Care Possible in Medical Desertsalmost 4 years ago
Women in Pharmacy Make Their Mark on the Professionalmost 4 years ago
Flip the Pharmacy Transforms Community Practice, One Pharmacy at a Timealmost 4 years ago
New Highs in Cannabis Pharmacyalmost 4 years ago
Care at the Counter: Women's Health in the PharmacyNewsletter
Pharmacy practice is always changing. Stay ahead of the curve with the Drug Topics newsletter and get the latest drug information, industry trends, and patient care tips.