Respiratory
Latest News
Latest Videos
CME Content
More News

A recent study reveals COVID-19 vaccination significantly reduces severe maternal disease and preterm birth risks during pregnancy.

Discover critical insights on respiratory health, COVID-19 impacts, and vaccination strategies from recent studies and regional policy changes.

A pharmacist's guidance is essential when selecting effective OTC cold and influenza medications that provide relief from symptoms such as cough, congestion, and fever.
Megan Smith, PharmD, discusses how pharmacists have taken reigns in boosting pneumococcal vaccine rates across health care.

Pharmacists enhance asthma care by bridging knowledge gaps, improving medication adherence, and providing cost-effective treatment options for patients.
Vaccine hesitancy rises as new recommendations emerge, highlighting pharmacists’ crucial role in addressing misinformation and improving immunization rates.

Multifaceted antimicrobial stewardship initiatives significantly enhance antibiotic prescribing practices for respiratory infections, improving patient outcomes.

Dietary nutrients show promise in reducing asthma risk linked to indoor air pollution, offering new strategies for pharmacists in patient care.

A recent review highlights cardiovascular risks linked to albuterol use in asthma treatment, urging safer alternatives and enhanced patient monitoring strategies.
With disease cases increasing substantially between 2012 and 2024, higher-valent vaccines are necessary to protect against nonvaccine serotypes and invasive pneumococcal disease.
A study shows dupilumab significantly improves symptoms of allergic fungal rhinosinusitis, offering hope for effective treatment options.
Study findings highlight the need for further exploration into the racial and ethnic trends of pneumococcal vaccination rates.
A study reveals a significant decline in inhaled corticosteroid use among COPD patients, highlighting the need for careful management and monitoring.
Georgia Travlos, PharmD, discusses changes in influenza and respiratory syncytial virus immunizations and coadministration best practices.

Tezspire gains FDA approval as the first biologic for chronic rhinosinusitis with nasal polyps, offering improved patient outcomes.

The approval expands options for protecting infants from respiratory syncytial virus, which remains a leading cause of pediatric hospitalization.
A recent study reveals a heightened stroke risk in asthma patients recovering from severe COVID-19, emphasizing the need for proactive monitoring and management.

Pharmacists can help to address access issues, offer comprehensive medication counseling, and educate patients about disease management.
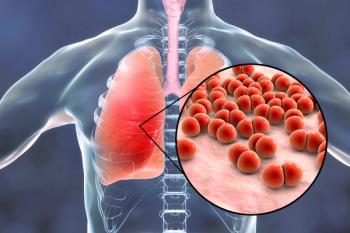
The study results highlight the importance of pneumococcal vaccination for patients with chronic respiratory diseases, particularly chronic obstructive pulmonary disease.

As their scope of practice continues to expand, researchers issue a survey on how community pharmacists conduct point-of-care testing for acute upper respiratory tract infections.
The recommendations are provided through the West Coast Health Alliance and pull from guidance from the American Academy of Pediatrics and other organizations.

As Walgreens senior director of immunizations, Samantha Picking, PharmD, delved into the evolution of pharmacies’ immunization services as well as their dedication to vaccine uptake.
Pharmacies play a significant role in influenza vaccination.

Researchers conducted a review of pharmacist interventions and their ability to impact the outcomes of patients with COPD.

Researchers determined the impact COVID-19 interventions had on Danish children’s incidence of RSV, influenza, and invasive pneumococcal disease.