Lauren Biscaldi, MS
Articles by Lauren Biscaldi, MS


AstraZeneca is the second pharmaceutical company to cap out-of-pocket costs for eligible patients.
Study results demonstrated the efficacy of semaglutide (Wegovy) in reducing cardiovascular risks in adults with overweight or obesity without diabetes.
This is the fifth oncology indication for zanubrutinib in the United States.

The out-of-pocket price cap applies to all inhalers in the company’s portfolio and will launch on June 1, 2024.

Dexcom's Stelo will be available in summer 2024.

The agency approved denosumab-bddz under two brand names: Wyost and Jubbonti.
The FDA granted accelerated approval to amivantamab-vmjw in May 2021.

The decision comes a year after a group of conservative attorneys generals threatened the chains with legal action.
Pfizer's RSV vaccine received FDA approval on May 31, 2023.

Three times each year, the National Community Pharmacists Association holds intensive workshops that drill down into the nitty gritty of pharmacy ownership.
In order to provide the best care, it’s crucial that pharmacists stay up-to-date on the latest guidance and data for flu, COVID-19, and RSV—even when that information is constantly changing.


These OTC products are recommended to address a variety of symptoms that occur during cold and flu seasons.

The approval was based on positive data from a phase 3 trial that showed patients treated with omalizumab were significantly more likely to tolerate peanut, milk, egg, or cashew protein without moderate to severe allergic symptoms.

According to Tyler Milliken, educating providers about prior authorization, along with other insurance processes and policies, plays a crucial role in advancing patients along their path to care.

In this conversation from the Healthcare Advocate Summit, Ned Woody, associate director of field reimbursement at Daiichi Sankyo Inc. - Oncology, discusses the disruptive impact that these programs have on patients.

Tyler Woods, PharmD, was joined by Ajay Mehra, chief financial officer and cofounder of UGO Rx, and Scott Cady, PharmD, during the Total Pharmacy Solutions Summit Fall 2023 to discuss how pharmacists can maximize opportunities to improve their stores.

Advocates can amplify the voices of transgender patients as they overcome barriers to health care, said Molly Crowther in a conversation at the 2023 Healthcare Advocate Summit.


Biologic approvals ranged from gene therapies for hemophilia A to a cellular therapy to treat type 1 diabetes.

In addition to the numerous adalimumab (Humira) biosimilars that launched this year, multiple new biosimilar products were approved by the FDA in 2023.
Vaccine approvals included two immunizations for RSV and a new pentavalent meningococcal shot.
Immunizations against influenza, COVID-19, and RSV are lower than they were this time last year, and hospitalizations have increased across all age groups.

Data from the Surveillance, Epidemiology, and End Results (SEER) database were reviewed.
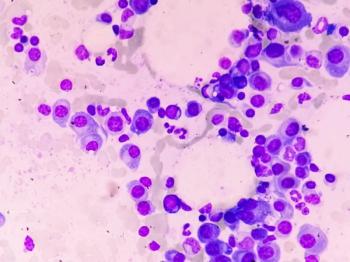
Understanding the adverse event profile of bispecific antibodies can aid treatment selection and improve patient outcomes.
