In the Opioid Epidemic, Treatment Challenges Still Persist
Despite societal need, stigma around MAT and MOUD still exists.
When the opioid epidemic began in the mid-1990s, it was impossible to predict just how many lives would be touched by addiction. By 2021, the number of people who died from a drug overdose was more than 6 times higher than it had been in 1999, and more than 75% of the 107,000 drug overdose deaths recorded in 2021 involved an opioid.1
In the decades since the epidemic took hold, pharmacists have grown more and more involved in treatment for opioid use disorder, providing counseling, medications, and other forms of treatment to individuals with opioid use disorder within their communities.
There are challenges in the system, said Thomas Menighan, MBA, ScD, FAPhA, CEO Emeritus of the American Pharmacists Association. “[Individuals with addiction] face a system that says, ‘Okay, this is not a moral failing to have an addiction.’ But we treat these folks like criminals, and that’s a conundrum we’ve got to try to solve, and part of what we’re here to talk about today.”
Menighan was joined on stage by Mark Garofoli, PharmD, MBA, BCGP, CPE, CTTS, director of experiential learning and a clinical assistant professor at the West Virginia University School of Pharmacy, and Tyler Varisco, PharmD, PhD, an assistant professor at the University of Houston College of Pharmacy, at the National Community Pharmacists Association 2023 Annual Convention and Expo.2
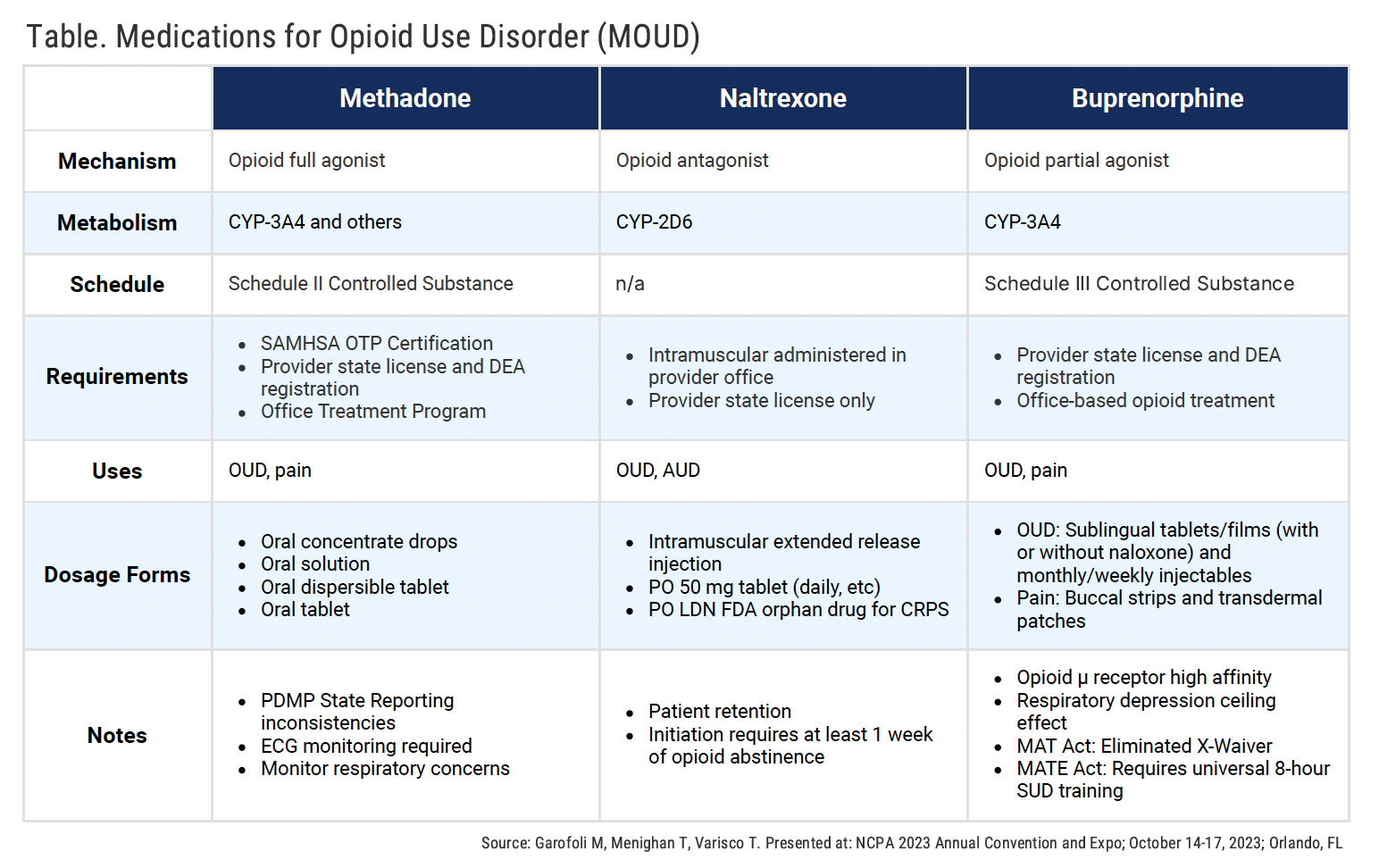
Medication Treatment Options
“Truth be told, we could all be in here for 4- or 8-hour days and days, talking about this,” said Garofoli. “Terminology is really important. There’s MAT, there’s MOUD… We’re all over the place.”
Treatment Access Issues Abound
While treatment options are available, it can be challenging to get MAT and MOUD therapies to patients who need it.
“I’m not here to throw any of the parties involved under the bus,” said Menighan. “There are a lot of well-meaning people out there, in all sectors: the regulatory sector, and the distribution sector. All of us have our rules and regulations that we have to follow; we have our settlement agreements [that] we have to meet the terms of.”
Currently, only 49% of pharmacies in the United States can dispense a 7-day supply of buprenorphine, and within the first 180 days of treatment, 1 in 3 patients will have to change pharmacies to continue accessing their medication. And frequently, those patients face stigma and concerns that they are simply “trading addictions,” from opioids to the medications used to treat them.
“You’re all aware of the red flags in access to buprenorphine, and your prescription drug monitoring programs all have red flags,” said Menighan, to help pharmacists identify patients who might be doctor shopping, bouncing around to different pharmacies, or taking multiple medications and prescriptions. “I’m a fan of what I’d characterize as green flags. And the green flags should matter just as much as the red flags.”
For Menighan, these green flags “are things like, is the patient staying on their therapy? You might, in a conversation with them, learn that they have a job now, and they’re keeping that job. Are they seeing their behavioral health specialist and getting the right results on their toxicology screens? All of those things should count for something, and we should be methodical and thoughtful about documenting those things in our records.”
Despite the focus on these green flags, there are still significant barriers in place for pharmacies that stock and distribute these medications. Pharmacies may now be able to document their NarxCare Score—a number ranging from 000 to 999 that reflects previous patient exposure to narcotics, sedatives, and stimulants, “with higher scores representing a relatively higher risk of overdose or potential misuse”3—Mengihan pointed out that what he as a pharmacist sees as a reasonable score is not considered reasonable to his wholesalers.
“If we’re ever challenged about our dispensing practices, we could say, ‘Well our average patient’s NarxCare Score is 300,’ which is reasonable; that should count for something. The challenge is, today, it doesn’t,” he explained. “All the wholesalers are able to look at the buprenorphine to suboxone ratio, and the control to non-control ratio, and if your pharmacy is an average, you’ve got a problem.”
A discussion with the wholesaler will likely ensue, he added, and the compliance team will take over. “I’ve had multiple conversations with all of the major wholesalers, and many of the regional and smaller ones, and it’s a challenge for them,” Mengihan said. “While they may be sympathetic to the need, to the societal need, for buprenorphine, at the end of the day, they’re beholden to settlement agreements—not with the DEA, who is…saying the right things, and trying to move toward a more supportive opportunity for buprenorphine in the market.”
But, he continued, “the settlement agreements aren’t with the DEA. They’re with the Justice Department.” And because the Department of Justice sits above the DEA, “until we can get the DOJ to say, ‘Yeah, we agree, we need to have more open access to buprenorphine,’ we still have challenges.”
Reference
Understanding the epidemic. CDC. Reviewed August 8, 2023. Accessed October 20, 2023. https://www.cdc.gov/opioids/basics/epidemic.html
Garofoli M, Menighan T, Varisco T. Pharmacy solutions for opioid use disorder: MOUD and stigma reduction. Presented at: National Community Pharmacists Association 2023 Annual Convention and Expo; October 14-17, 2023; Orlando, FL.
NarxCare Scores — a refresher. Iowa Board of Pharmacy. March 2023. Accessed October 20, 2023. https://nabp.pharmacy/wp-content/uploads/2023/03/March-2023-Iowa-State-Newsletter.pdf

Q&A: Pharmacist-Led Diabetes Management Transforms Patient Care and Clinical Outcomes
June 28th 2025An ambulatory care pharmacist program in Washington State dramatically improved diabetes management by reducing A1c levels, increasing medication adherence, and providing comprehensive patient education.