Melphalan flufenamide (Pepaxto; Oncopeptides AB) in combination with dexamethasone was granted accelerated approval on February 26, 2021, by the FDA for adult patients with relapsed or refractory multiple myeloma.
Associate Clinical Professor, University of Connecticut School of Pharmacy, Storrs, CT.

Melphalan flufenamide (Pepaxto; Oncopeptides AB) in combination with dexamethasone was granted accelerated approval on February 26, 2021, by the FDA for adult patients with relapsed or refractory multiple myeloma.

Belantamab mafodotin-blmf is now indicated for adult patients with relapsed or refractory myeloma who have received at least 4 prior therapies.
On October 22, 2020, the FDA approved remdesivir (Veklury; Gilead) as the first treatment for coronavirus disease 2019 (COVID-19).

The FDA approved azacitidine tablets (Onureg; Celgene Corporation) on September 1, 2020
On May 8, 2020, the FDA granted accelerated approval to selpercatinib (Retevmo; Eli Lilly and Company).
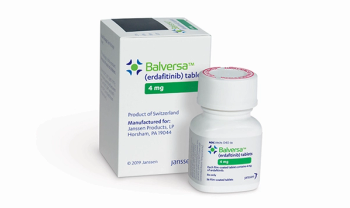
First oral drug for advanced or metastatic bladder cancer.

Both responder and remitter patients experienced a statistically significant longer time to relapse with esketamine compared to the placebo
FDA approves apalutamide for castration-resistant prostate cancer.
Emicizumab-kxwh brings together factors IXa and X to restore the function of the missing activated factor VIII and the blood clotting process.

A new era of treatment has arrived-here’s what you need to know.

The New Drug Review for April

Published: December 4th 2020 | Updated:

Published: February 12th 2021 | Updated:
Published: July 22nd 2020 | Updated:
Published: January 14th 2021 | Updated:

Published: November 9th 2017 | Updated:

Published: April 17th 2017 | Updated: