
Check out important updates from the FDA for the week of August 5.

Sam Clark, MD, PhD, founder and CEO of Terran Biosciences, discusses the implications of a potential FDA approval of MDMA-assisted therapy for PTSD.

In a clinical trial, IPX203 was able to achieve longer good on-time with less doses compared to standard treatment.

Nalmefene injection is an opioid receptor antagonist and comes in a single-dose, pre-filled auto-injector that delivers 1.5 mg of the medication per actuation.

Check out important updated from the FDA for the week of July 29.

Afami-cel is the first new treatment for synovial sarcoma in more than 10 years, ending a long-standing therapeutic gap.

Pharmacists should be knowledgeable about the symptoms of chemical peel irritation to effectively advise patients.

In 3 Phase 3 trials, elinzanetant reduced the frequency and severity of moderate to severe vasomotor symptoms, and improved sleep disturbances.

The investigational oral, selective NaV1.8 pain signal inhibitor from Vertex Pharmaceuticals demonstrated a favorable benefit/risk profile in 3 phase 3 trials.

The new FDA-approved presentation will be the company’s third 505(b)(2) injectable added in 2024, expected to launch in the third quarter.

Check out important updates from the FDA from over the past week.

The approval of eculizumab-aagh offers more treatment options for patients with rare diseases PNH and aHUS.

The approval offers a first of its kind treatment option for patients living with nonerosive GERD.

The companies are marketing products that may violate Federal Trade Commission prohibition of “unfair and deceptive acts in the marketplace.”

Had Novo Nordisk’s insulin icodec secured FDA approval, it would have been the first and only once-weekly basal insulin option for adults with diabetes.

The commercial product is expected to be available by the end of July.
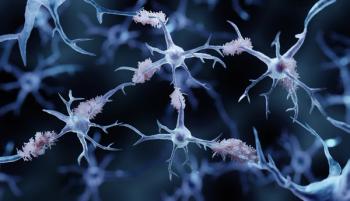
Donanemab-azbt was approved under the FDA's Fast Track Review, Priority Review, and Breakthrough Therapy designations.
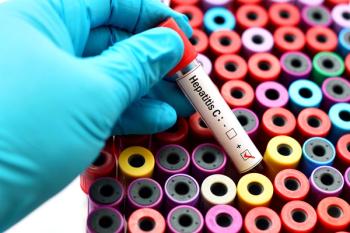
Through a test-and-treat approach, patients who test positive for HCV RNA with the Xpert HCV test and GeneXpert Xpress System can be connected to care as soon as the same health care visit.

Check out these important FDA updates from the month of June 2024.

The first-in-class selective dual PDE3 and PDE4 inhibitor from Verona Pharma is the first inhaled product with a novel mechanism of action approved to treat COPD in over 20 years.
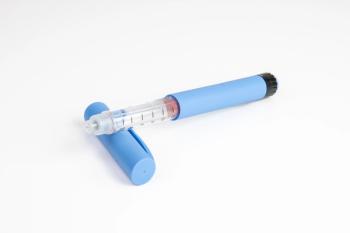
The Victoza generic is approved for use in adults and children aged 10 years and older.

Check out important updates from the FDA for the week of June 17.

Cefepime/enmetazobactam was granted a 5-year exclusivity extension under GAIN Act legislation.
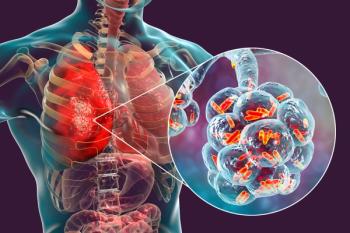
The vaccine covers 8 serotypes not currently covered by any FDA-approved pneumococcal vaccines.

The therapy from AstraZeneca is indicated to improve glycemic control in patients aged 10 years or older.

The Lingo and Libre Rio from Abbott are based on the company’s FreeStyle Libre sensing technology.

Sam Clark, MD, PhD, founder and CEO of Terran Biosciences, discusses how some of the concern’s raised by the panel can inform future psychedelic research.

The new task force highlights a stronger government focus on curbing the risks of e-cigarettes for young people, especially after sustained calls for stricter action on the public health issue by lawmakers and the public.

Sam Clark, MD, PhD, founder and CEO of Terran Biosciences, talks about some of the problems he saw during the Psychopharmacologic Drugs Advisory Committee meeting.